
Figure 1 (left). Potassium (K) deficiency in Cabernet Franc. Note leaves turning red in between the vein.
Figure 2 (center). Magnesium (Mg) deficiency in Chambourcin. Note wedges of discoloration, starting from edges.
Figure 3 (right). Phosphorous (P) deficiency in Zinfandel. Note the reddening progression from the leaf margins.
Fact Sheet FS1260

Figure 4. Grapevine leafroll disease symptoms on Cabernet Sauvignon. Note the reddish discoloration on interveinal areas of the leaf, but the veins stay green and the downward cupping or curling from the edges.
Red leaves in the vineyard are caused by many biotic (viruses, bacteria, and fungus) and abiotic (nutrient deficiencies, cold injury, damage to root systems, etc.) stresses. Anything that can cause blockage or stress in the vascular system where water and nutrients are transported can result in the development of red leaves. Though the timing, pattern of appearance, and spread could be different, the overlapping of the symptoms, especially the reddening of leaves, makes it very difficult to identify the cause based only on visual symptoms.1 The best strategy therefore is not to rely on guesswork, but to get the vines tested for accurate diagnosis as soon as possible.
Below are examples of abiotic and biotic stresses that are often associated with red leaves on grapevines.
Nutrient Deficiencies
Potassium (K) deficiency develops when vines receive less K than what is required for normal growth and development, or with low soil pH levels (i.e., less than 5 for Vitis vinifera) that decrease the availability of potassium to the roots. Potassium is a mobile nutrient and in the case of deficiency, the upper (younger) leaves receive K rather than the lower (older) leaves, therefore reddening the leaves in the lower part of the canopy first.
Symptoms typically appear in early to late summer.2 In red grape varieties, leaves turn red in-between the veins starting from the edges, and as deficiency gets severe, they turn purple (Figure 1). In white grape varieties, deficient leaves turn chlorotic (pale yellow or white), due to insufficient chlorophyll.

Figure 5. Grapevine leafroll disease symptoms on Chardonnay. Note the interveinal area turning pale green while the veins turn yellowish and the downward cupping or curling from the edges.
To monitor plant K status, it is advisable to perform petiole analysis every 2–3 years, ideally during bloom. The target K level in petioles should be between 1.5% to 2.5% if sampled during bloom, and 1.2% to 2.0% if sampled 70 days after bloom. Apply potash (KOH) fertilizer either foliar or to the soil, depending on the urgency. When there is chronic potassium deficiency, laboratory soil testing for nutrient and pH levels should be conducted.
Magnesium (Mg) deficient leaves may resemble K deficient leaves; however the discoloration starts from the end, creating the wedge shaped pattern (Figure 2). As the deficiency becomes severe in red varieties, interveinal chlorosis could become red to brown. In white varieties, the chlorosis remains yellowish. Target Mg level in petioles should be 0.3% to 0.5% at bloom; or between 0.35% to 0.75% if sampled 70 days after bloom.2 To correct the Mg deficiency, magnesium salt (MgSO4), also known as Epsom salts, is used if soil pH does not require adjustments. If your soil is acidic, apply dolomitic limestone (Mg-containing limestone) as it will also raise the pH, improving the rate of Mg uptake.

Figure 6. Grape mealybugs (Pseudococcus maritimus) on a grape shoot.
Phosphorous (P) deficient leaves show interveinal reddening while white varieties show chlorosis.2 Typically, initial chlorosis appears around leaf margins, which turn reddish in red varieties (Figure 3). Unlike other major nutrients, P offers greater flexibility in terms of timing of application as it is less mobile in soil and does not leech away easily. The target P level for leaf petiole should be between 0.13 to 0.30 % depending on the type of rootstock. Mono-ammonium phosphate (MAP) and di-ammonium phosphate (DAP) are common sources of phosphorous fertilizer.2 Adjust the soil pH if it is lower than optimum to improve P uptake.
Diseases
Viruses of grapes such as leaf roll and red blotch can also cause red leaves and result in reduced vine vigor, poor fruit set, reduced fruit quality, and early decline of vines. Since there is no cure available for viruses once vines are infected, the only way to cure the infected vineyard is rogueing out infected vines, and replanting with virus-tested planting materials from dependable nurseries.3

Figure 7. Grapevine red blotch disease symptoms on Merlot. Note red pigment appearing randomly on leaf. Reddening from edges can be mistaken for K deficiency and rolling or curling at edge can be mistaken for leaf roll virus symptoms.
Grapevine leafroll disease symptoms typically appear around the onset of fruit ripening and spread as the season progresses. In red varieties, red to purplish discoloration is observed on interveinal areas of the leaf, but the veins stay green (Figure 4). In white varieties, the interveinal area will become pale green while the veins turn yellowish (Figure 5). In addition, downward cupping or curling from the edges is typical of leafroll symptoms (Figure 5). The discoloration of leaves will often develop randomly within the vine canopy. An upward or downward movement of the discoloration will not occur as it does with some of the nutrient deficiencies.
There are several viruses that can cause grapevine leafroll disease, and grapevine leafroll-associated virus-1 and -3 (GLRaV-1 and -3) can be disseminated by mealybugs or scale insects (Figure 6). Scouting for these vector insects can be done during the season (around bloom) by carefully examining the basal area of shoots and underneath the bark. Mealybugs can be managed through insecticide treatment(s), however recent research showed that even a small population of mealybugs could quickly spread grapevine leafroll disease.

Figure 8. Crown gall symptom at the graft union.
Grapevine red blotch disease is a recently recognized virus that has existed for a long time. As the name suggests, blotches of red pigment appear randomly on leaves of infected vines. It can be detected at any stage of vine growth in any part of a vine. It can easily be mistaken for potassium deficiency or leaf roll virus, especially when leaves cup or roll (Figure 7). Because of the recent recognition of the virus, there are ongoing efforts to understand how (or if) it spreads in vineyards. It is known that the red blotch virus can be disseminated through propagation and grafting with infected materials.
For the two viruses mentioned above and any other viruses, the vines will always be infected. Thus, it is recommended to obtain virus-tested materials from reputable nurseries when establishing a vineyard. Confirm that the nursery has virus-testing procedures in place such as Protocol 2010.4 The materials from Protocol 2010 have been available since 2013, and availability has increased as more nurseries have adapted the protocol.
If you have propagated your planting materials from existing vineyards, or obtained materials from non-certified nurseries, consider sending a representative number of cuttings for virus-testing before planting. Typically, 7–8 young, matured leaves per vine, and 30–50 leaves per sample are sufficient, but communicate with the virus-testing lab (listed below) for their specific sampling protocol. When one of the vector insects and one of the grapevine leafroll-associated viruses (GRLaV-1 or -3) are present in your vineyard, management of the vector insect will be required to keep healthy vines clean of GLRaV.

Figure 9. A series of small crown gall formed under the bark of Merlot.
Crown Gall is caused by a bacterium Rhizobium ( = Agrobacterium) vitis and can be common in cool climates. Severe winters or temperature fluctuations in the early spring damage bark and vascular tissues and allow R. vitis to move into the vine cells and cause infection.5 Typical symptoms are formation of galls on the trunk (Figure 8); however, these galls can be very small, and may be formed under the bark (Figure 9). Therefore, crown gall could be mistakenly identified as a nutrient deficiency, as red leaves resulting from the stressed vascular system are readily visible (Figure 10). Other wounding events, such as physical damage to the trunk by hitting it with equipment such as line trimmers, can also encourage the infection by R. vitis.
As with virus diseases, crown gall-infected vines are infected for life because crown gall bacterium is a systemic pathogen. Thus, management of crown gall is very difficult. Obtain clean plant materials (note: the procedure used in Protocol 2010 should considerably reduce the risk of R. vitis from the mother plant), and make sure to select varieties that suit your site. Cold tender varieties, such as Merlot and Tanat tend to have higher risk of crown gall than many others do. Moreover, stressed vines tend to be more cold tender, thus, proper viticultural practices such as site selection, crop load, and foliar disease management can lower the risk of crown gall. Having multiple trunks from above the graft union and hilling up to cover the graft unions during the winter months are other preventative strategies.
Assessment of Suspicious Vines
It can be very difficult to accurately identify the cause of red leaves. Virus testing (for leafroll, red blotch, and others) is expensive, hence the first recommendation in an identification flowchart is to inspect trunks for integrity, and then perform petiole analysis (Figure 11). If trunk diseases and nutrient deficiencies are ruled out, samples should be sent for virus testing. Below is a list of grapevine virus testing laboratories. Lab testing pricing varies, so make sure to contact several companies for cost comparison.

Figure 10. Red foliar discoloration caused by crown gall. Note the gall formation on the trunk.
Virus Testing Labs
-
Agri-Analysis LLC
930 Riverside Parkway, Suite #30, West Sacramento, CA 95605
Phone: 800-506-9852
Email: info@agri-analysis.com -
AL&L Crop Solutions
7769 N. Meridian Rd., Vacaville, CA 95688
Phone: 530-759-9460 Email:
info@allcropsolutions.com -
Eurofins STA Laboratories
7240 Holsclaw Road, Gilroy, CA 95020
Phone: 888-782-5220
Email: juditmonis@eurofinus.com -
Sunburst Plant Disease
Clinic 677 East Oliver Avenue, Turlock, CA 95380
Phone: 209-667-4442
Email: slivingston@sunburstpdcinc.com -
WSU ELISA Virus Testing Lab
24106 N. Bunn Rd., Prosser, WA 99350
Phone: 509-786-9382
Email: Tina_Vasile@wsu.edu
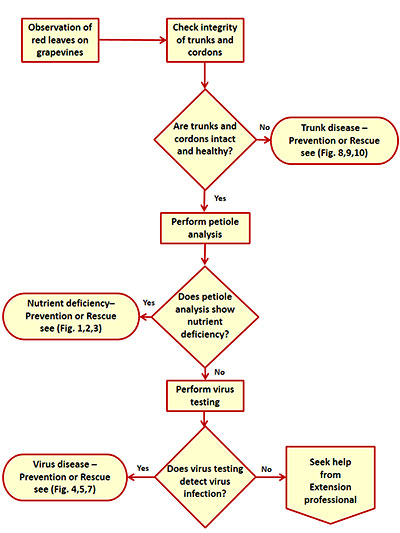
Figure 11. A flowchart for actions to be taken after you observed red leaves on grapevines.
Cited References
Photo Credits
- Photos for Figures 1, 2 and 3 by Hemant Gohil
- Photos for Figures 4, 5, 6, 8, 9 and 10 by Mizuho Nita
- Photo for Figure 7 by Prashant Swamy
Mention or display of a trademark, proprietary product, or firm in text or figures does not constitute an endorsement by Rutgers Cooperative Extension and does not imply approval to the exclusion of other suitable products or firms.
October 2016
Copyright © 2024 Rutgers, The State University of New Jersey. All rights reserved.
For more information: njaes.rutgers.edu.
Cooperating Agencies: Rutgers, The State University of New Jersey, U.S. Department of Agriculture, and Boards of County Commissioners. Rutgers Cooperative Extension, a unit of the Rutgers New Jersey Agricultural Experiment Station, is an equal opportunity program provider and employer.



